|
Suppliers for CAS
123171-59-5
|
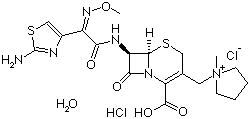
Properties | | CAS |
123171-59-5 | | Formula |
C19H24N6O5S2 . 2HCl |
|
|
19 Registered suppliers
Dayang Chem (Hangzhou) Co.,Ltd.
P.R.China
Hangzhou Meite Industry Co., Ltd (Hangzhou Meite Chemical Co., Ltd)
P.R.China
Leap Chem Co., Ltd
P.R.China
Molecular Formula: C19H24N6O5S2.2ClH.H2O Molecular Weight: 571.5 Hazard Symbols: Xi WGKGermany: 3 HS Code: 29419000
More details are to be found here
X´ian Taicheng Chem Co., Ltd
P.R.China
Molecular Formula: C19H24N6O5S2.2ClH.H2O Molecular Weight: 571.5 Hazard Symbols: Xi WGKGermany: 3 HS Code: 29419000
More details are to be found here
Hangzhou Zhongqi Chem Co., Ltd
P.R.China
Molecular Formula: C19H24N6O5S2.2ClH.H2O Molecular Weight: 571.5 Hazard Symbols: Xi WGKGermany: 3 HS Code: 29419000
More details are to be found here
Nantong Xindao Biotech Ltd
P.R.China
Molecular Formula: C19H24N6O5S2.2ClH.H2O Molecular Weight: 571.5 Hazard Symbols: Xi WGKGermany: 3 HS Code: 29419000
More details are to be found here
Hangzhou Lingrui Chemical Co., Ltd.
P.R.China
Molecular Formula: C19H24N6O5S2.2ClH.H2O Molecular Weight: 571.5 Hazard Symbols: Xi WGKGermany: 3 HS Code: 29419000
More details are to be found here
Skyrun Industrial Co., Ltd.
P.R.China
Molecular Formula: C19H24N6O5S2.2ClH.H2O Molecular Weight: 571.5 Hazard Symbols: Xi WGKGermany: 3 HS Code: 29419000
More details are to be found here
BOC Sciences
USA
Description : Cefepime is an extended-spectrum parenteral cephalosporin antibiotic active in vitro against a broad spectrum of gram-positive and gram-negative aerobic bacteria. Cefepime dosing was 1-4 g/day (0.5-2.0 g twice daily) for adults; ceftazidime dosing was 1-6 g/day (0.5 g every 12 hours to 2.0 g every 8 hours). A limited number of cefepime-treated patients received 2 g every 8 hours. The median length of dosing for both cefepime and ceftazidime was 7 days. Cefepime has a decreased propensity to induce beta-lactamases compared with other beta-lactam antibiotics. Cefepime has a pharmacokinetic disposition similar to that of other renally eliminated cephalosporins, with a half-life of approximately 2 hours. Cefepime has demonstrated clinical efficacy against a variety of infections, including urinary tract infections, pneumonia, and skin structure infections. Cefepime is generally well tolerated. - Molecular Weight :571.50
- Melting Point :150°C(dec.)
- Purity :> 98%
Molecular Formula : C19H28Cl2N6O6S2 Canonical SMILES : C[N+]1(CCCC1)CC2=C(N3C(C(C3=O)NC(=O)C(=NOC)C4=CSC(=N4)N)SC2)C(=O)O.O.Cl.[Cl-]
InChI :
InChI=1S/C19H24N6O5S2.2ClH.H2O/c1-25(5-3-4-6-25)7-10-8-31-17-13(16(27)24(17)14(10)18(28)29)22-15(26)12(23-30-2)11-9-32-19(20)21-11;;;/h9,13,17H,3-8H2,1-2H3,(H3-,20,21,22,26,28,29);2*1H;1H2/b23-12-;;;/t13-,17-;;;/m1./s1
InChIKey :
LRAJHPGSGBRUJN-OMIVUECESA-N
Solubility :
Soluble in DMSO
Appearance :
White Solid
Storage : Store at -20°C
Synonyms :
Maxipime
More details are to be found here
Shandong SanYoung Industry Co., Ltd
P.R.China
Description : Cefepime is an extended-spectrum parenteral cephalosporin antibiotic active in vitro against a broad spectrum of gram-positive and gram-negative aerobic bacteria. Cefepime dosing was 1-4 g/day (0.5-2.0 g twice daily) for adults; ceftazidime dosing was 1-6 g/day (0.5 g every 12 hours to 2.0 g every 8 hours). A limited number of cefepime-treated patients received 2 g every 8 hours. The median length of dosing for both cefepime and ceftazidime was 7 days. Cefepime has a decreased propensity to induce beta-lactamases compared with other beta-lactam antibiotics. Cefepime has a pharmacokinetic disposition similar to that of other renally eliminated cephalosporins, with a half-life of approximately 2 hours. Cefepime has demonstrated clinical efficacy against a variety of infections, including urinary tract infections, pneumonia, and skin structure infections. Cefepime is generally well tolerated. - Molecular Weight :571.50
- Melting Point :150°C(dec.)
- Purity :> 98%
Molecular Formula : C19H28Cl2N6O6S2 Canonical SMILES : C[N+]1(CCCC1)CC2=C(N3C(C(C3=O)NC(=O)C(=NOC)C4=CSC(=N4)N)SC2)C(=O)O.O.Cl.[Cl-]
InChI :
InChI=1S/C19H24N6O5S2.2ClH.H2O/c1-25(5-3-4-6-25)7-10-8-31-17-13(16(27)24(17)14(10)18(28)29)22-15(26)12(23-30-2)11-9-32-19(20)21-11;;;/h9,13,17H,3-8H2,1-2H3,(H3-,20,21,22,26,28,29);2*1H;1H2/b23-12-;;;/t13-,17-;;;/m1./s1
InChIKey :
LRAJHPGSGBRUJN-OMIVUECESA-N
Solubility :
Soluble in DMSO
Appearance :
White Solid
Storage : Store at -20°C
Synonyms :
Maxipime
More details are to be found here
Carbone Scientific Co., Ltd.
U.K.
Description : Cefepime is an extended-spectrum parenteral cephalosporin antibiotic active in vitro against a broad spectrum of gram-positive and gram-negative aerobic bacteria. Cefepime dosing was 1-4 g/day (0.5-2.0 g twice daily) for adults; ceftazidime dosing was 1-6 g/day (0.5 g every 12 hours to 2.0 g every 8 hours). A limited number of cefepime-treated patients received 2 g every 8 hours. The median length of dosing for both cefepime and ceftazidime was 7 days. Cefepime has a decreased propensity to induce beta-lactamases compared with other beta-lactam antibiotics. Cefepime has a pharmacokinetic disposition similar to that of other renally eliminated cephalosporins, with a half-life of approximately 2 hours. Cefepime has demonstrated clinical efficacy against a variety of infections, including urinary tract infections, pneumonia, and skin structure infections. Cefepime is generally well tolerated. - Molecular Weight :571.50
- Melting Point :150°C(dec.)
- Purity :> 98%
Molecular Formula : C19H28Cl2N6O6S2 Canonical SMILES : C[N+]1(CCCC1)CC2=C(N3C(C(C3=O)NC(=O)C(=NOC)C4=CSC(=N4)N)SC2)C(=O)O.O.Cl.[Cl-]
InChI :
InChI=1S/C19H24N6O5S2.2ClH.H2O/c1-25(5-3-4-6-25)7-10-8-31-17-13(16(27)24(17)14(10)18(28)29)22-15(26)12(23-30-2)11-9-32-19(20)21-11;;;/h9,13,17H,3-8H2,1-2H3,(H3-,20,21,22,26,28,29);2*1H;1H2/b23-12-;;;/t13-,17-;;;/m1./s1
InChIKey :
LRAJHPGSGBRUJN-OMIVUECESA-N
Solubility :
Soluble in DMSO
Appearance :
White Solid
Storage : Store at -20°C
Synonyms :
Maxipime
More details are to be found here
Xiamen Equation Chemical Co.,Ltd
P.R.China
Description : Cefepime is an extended-spectrum parenteral cephalosporin antibiotic active in vitro against a broad spectrum of gram-positive and gram-negative aerobic bacteria. Cefepime dosing was 1-4 g/day (0.5-2.0 g twice daily) for adults; ceftazidime dosing was 1-6 g/day (0.5 g every 12 hours to 2.0 g every 8 hours). A limited number of cefepime-treated patients received 2 g every 8 hours. The median length of dosing for both cefepime and ceftazidime was 7 days. Cefepime has a decreased propensity to induce beta-lactamases compared with other beta-lactam antibiotics. Cefepime has a pharmacokinetic disposition similar to that of other renally eliminated cephalosporins, with a half-life of approximately 2 hours. Cefepime has demonstrated clinical efficacy against a variety of infections, including urinary tract infections, pneumonia, and skin structure infections. Cefepime is generally well tolerated. - Molecular Weight :571.50
- Melting Point :150°C(dec.)
- Purity :> 98%
Molecular Formula : C19H28Cl2N6O6S2 Canonical SMILES : C[N+]1(CCCC1)CC2=C(N3C(C(C3=O)NC(=O)C(=NOC)C4=CSC(=N4)N)SC2)C(=O)O.O.Cl.[Cl-]
InChI :
InChI=1S/C19H24N6O5S2.2ClH.H2O/c1-25(5-3-4-6-25)7-10-8-31-17-13(16(27)24(17)14(10)18(28)29)22-15(26)12(23-30-2)11-9-32-19(20)21-11;;;/h9,13,17H,3-8H2,1-2H3,(H3-,20,21,22,26,28,29);2*1H;1H2/b23-12-;;;/t13-,17-;;;/m1./s1
InChIKey :
LRAJHPGSGBRUJN-OMIVUECESA-N
Solubility :
Soluble in DMSO
Appearance :
White Solid
Storage : Store at -20°C
Synonyms :
Maxipime
More details are to be found here
AK Scientific, Inc.
USA
Description : Cefepime is an extended-spectrum parenteral cephalosporin antibiotic active in vitro against a broad spectrum of gram-positive and gram-negative aerobic bacteria. Cefepime dosing was 1-4 g/day (0.5-2.0 g twice daily) for adults; ceftazidime dosing was 1-6 g/day (0.5 g every 12 hours to 2.0 g every 8 hours). A limited number of cefepime-treated patients received 2 g every 8 hours. The median length of dosing for both cefepime and ceftazidime was 7 days. Cefepime has a decreased propensity to induce beta-lactamases compared with other beta-lactam antibiotics. Cefepime has a pharmacokinetic disposition similar to that of other renally eliminated cephalosporins, with a half-life of approximately 2 hours. Cefepime has demonstrated clinical efficacy against a variety of infections, including urinary tract infections, pneumonia, and skin structure infections. Cefepime is generally well tolerated. - Molecular Weight :571.50
- Melting Point :150°C(dec.)
- Purity :> 98%
Molecular Formula : C19H28Cl2N6O6S2 Canonical SMILES : C[N+]1(CCCC1)CC2=C(N3C(C(C3=O)NC(=O)C(=NOC)C4=CSC(=N4)N)SC2)C(=O)O.O.Cl.[Cl-]
InChI :
InChI=1S/C19H24N6O5S2.2ClH.H2O/c1-25(5-3-4-6-25)7-10-8-31-17-13(16(27)24(17)14(10)18(28)29)22-15(26)12(23-30-2)11-9-32-19(20)21-11;;;/h9,13,17H,3-8H2,1-2H3,(H3-,20,21,22,26,28,29);2*1H;1H2/b23-12-;;;/t13-,17-;;;/m1./s1
InChIKey :
LRAJHPGSGBRUJN-OMIVUECESA-N
Solubility :
Soluble in DMSO
Appearance :
White Solid
Storage : Store at -20°C
Synonyms :
Maxipime
More details are to be found here
Chemlyte Solutions
P.R.China
More details are to be found here
Chemos GmbH & Co. KG
Germany
More details are to be found here
BuGuCh & Partners
Germany
More details are to be found here
Sigma-Aldrich International GmbH
Switzerland
More details are to be found here
CM Fine Chemicals
Switzerland
More details are to be found here
Santa Cruz Biotechnology, Inc.
USA
More details are to be found here
|


 details
details details
details details
details_Co_Ltd_1_575.jpg)








